Modern Geology, 1993, Vol. 19, pp. 81-99 © 1993 Gordon and
Breach Science Publishers S.A.
Reprints available directly from the publisher Printed in Malaysia
Photocopying permitted by license only
| Note: In 2001 Brian Axsmith reported the discovery of a relatively complete speciment of Pannaulika triassica from the Solite quarry from where the leaf fragment described below came. He says the new specimen indicates an affinity with Clathropteris, which is a dipteridaceous fern. The new specimen indicates that P. triassica is a compound leaf, as Cornet predicted, but unless the new specimen is fertile (contains sori), its relationship to Clathropteris and ferns will depend on more than a superficial resemblance. For now Axsmith's interpretation needs to be considered when reading the paper below. Abstract at http://gsa.confex.com/gsa/2001SE/finalprogram/abstract_2670.htm |
DICOT -LIKE LEAF AND FLOWERS FROM THE
LATE TRIASSIC TROPICAL NEWARK
SUPERGROUP RIFT ZONE, U.S.A.
BRUCE CORNET
Lamont-Doherty Earth Observatory, Palisades, NY 10964
(Received 19 December 1992)
Pannaulika triassica Cornet, n. gen. et sp., a dicot-like leaf, is described from late Carnian lacustrine black shales of the Dan RiverjDanville basin of North Carolina and Virginia. Associated bicarpellate receptacle, achene, and multicarpellate spike are also described. The small simple pinnately-veined leaf has well-developed festooned brochidodromous secondary and tertiary veins, and a very ordered reticulate quarternary venation: Its asymmetry and torn margin imply that it is a fragment of a larger leaf, perhaps a lateral lobe of a palmately veined three-lobed leaf. A probable anthophyte affinity is based on comparisons with living and/or extinct ferns, pteridosperms, and anthophytes. It is considered to have had an upland tropical origin.
KEY WORDS: Late Triassic, angiosperm, dicot-like leaf, Pannaulika.
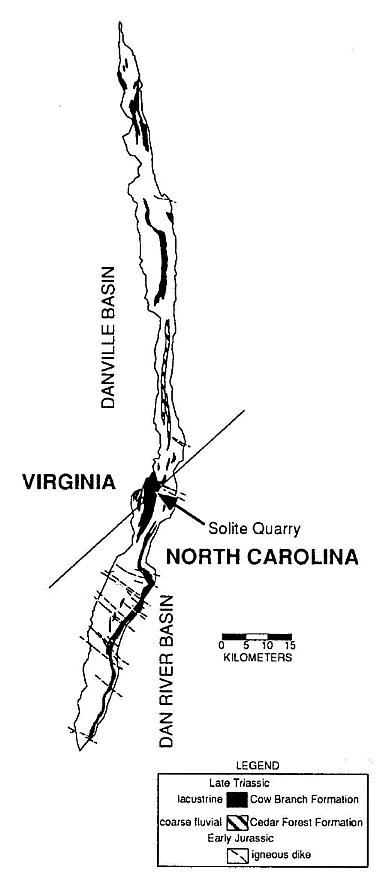
The fossil dicot-like leaf comesirom the Late Triassic Cow Branch Formation of the Dan River-Danville basin of North Carolina and Virginia, one of many Newark Supergroup rift basins in eastern North America (Olsen and Gore, 1989; Manspeizer and Cousminer, 1988; Cornet and Olsen, 1990; Olsen, 1990). It was discovered in a lacustrine, black, microlaminated calcareous claystone zone of the numerous shallow to deep lake level cycles exposed at the Solite quarry (Figures 1 and 2). A late Carnian age has been assigned based on lithostratigraphic and biostratigraphic correlation (Cornet and Olsen, 1985; Robbins-and Traverse, 1980; Robbins, 1982). More recently, a tropical paleogeographic position has been resolved by geomagnetic paleopole position for the Carnian (Witte et al., 1991), and by the analysis of the pattern of lake cycles exposed in the quarry that are a reflection of Milankovitch type orbitally-driven changes in solar insolation (Olsen, 1986). Because the periodicity of northern and southern hemisphere orbital precession cycles are out of phase, only near the equator can the effects of alternating orbital cycles of both hemispheres be detected. Lake cycles at the Solite quarry record prominent 10,000-13,000 yr. cycles, as well as the normal 18,000-24,000 yr. cycles (Figure 2), which supports paleomagnetic evidence of a paleoequatorial position (Olsen, 1991). It is with this possible source in the tropics and a late Carnian age in mind that the dicot-like leaf is described below.

Figure 1 Outline of the Dan River-Danville basin and the location of the Solite quarry, the source of the dicot-like leaf. Both the basin and the quarry straddle the Virginia-North Caronlina border.
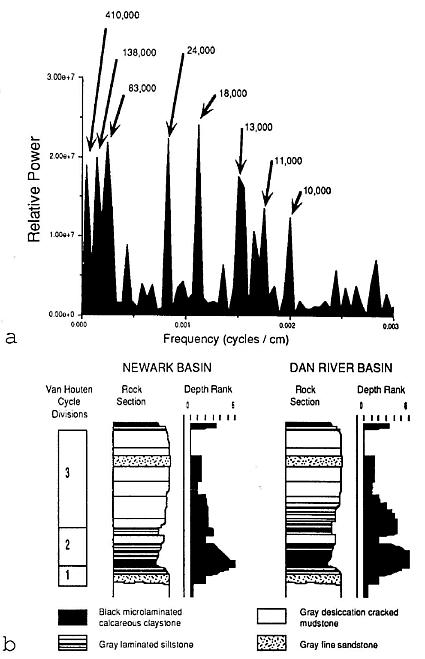
Figure 2 a, Power- spectrum measuring the relative frequency of sedimentary cycles perimeter in the Solite quarry..Fourier analysis was performed by P. E. Olsen on the cyclic sequence of depth ranks or paleoenvironmental facies through the twelve Van Houten cycles exposed in the quarry (Olsen, 1986; Olsen et al., 1978). Cycle duration in years for the various peaks is based on an average sedimentation rate for the 21,000 year precession cycle of 0.48 m per 1,000 years. b, Comparison of Van Houten cycles and their depth ranks in the Lockatong Formation (Newark basin) and the Cow branch Formation (Dan River basin). Note the double peaks formed by depth ranks greater than 3 in the Dan River basin, which was situated near the paleoequator at the time of deposition, but the presence of only one peak in the Newark basin, which is situated 200 km to the north of the Dan River basin.
SYSTEMATIC PALEOBOTANY
Pannaulika Cornet, new genus
Type species
Pannaulika triassica new and only known species.
Distribution
Currently same as for the species.
Diagnosis
Small leaf with entire margin and brochidodromous secondary veins arising excurrently from primary; tertiary veins form loops or arches outside secondary loops, and subaxially-oriented transverse veins occur within some of the secondary loops; very regular oriented quarternary reticulate venation developed inside secondary and tertiary loops; a band of dendritic or branching quarternary venation extends from tertiary loops or reticulum to the margin of leaf.
Etymology
Named after Paul E. Olsen and Annika Johansson. who discovered the part and counterpart of the fossil, respectively, on 17 May 1991. P-ann-aul-ika = anagram of PaulAnnika to give the following double meaning: Pan = Greek for the union of all branches of a group; aulik = a variation of aulic, Greek for royal court or place of authority or rule. The second more cryptic meaning is in reference to Axelrod's 1952 theory (Axelrod, 1952) of angiosperm origin in tropical uplands, i.e., the place that many hold the roots of the angiosperm phylogenetic tree or bush.
Remarks
Leaf incomplete, missing lower (proximal) margin on one side and lacking a base. Because this leaf may be a lobe or part of a larger leaf, the generic diagnosis does not address the overall shape and size of the leaf, nor does it address details about lamina and vein asymmetry, which at this time are considered aspects of the specific diagnosis.
Pannaulika triassica Cornet, new species (Figures 3d-g; 4a)
Type material
Holotype: VMNH201 (Figures 3e,f).
Repository
The Virginia Museum of Natural History, Martinsville, VA.
Distribution
Solite quarry, Late Triassic (late Carnian) Cow Branch Formation of southern Virginia and northern North Carolina, U.S.A. (Figure 1).
Figure 3 a, SEM enlargem.ent of exine of Monocrinopollis walkeri (Richmond basin, VB4, early Carnian), showing prominent columellae supporting a reticulum. b, Monocrinopollis doylei Comet (47 x 36 microns; Richmond basin, VB4, early Carnian). c, Monocrinopollis microreticulatus Comet (34 x 27 microns; 32 x 27 microns; Riclimond basin, VB4, early Carnian). d, Pannaulika triassica novo gen. et sp., photographed under high angle lighting with venation appearing silver-colored (x 4; late Carnian); a fossil conchostracan (Cyzicus) or clam shrimp is present in the upper left of photograph (bean-shaped reflection). e, Pannaulika triassica novo gen. et sp., entire specimen showing incomplete base and apex (x 2.2); veins represented by impressions in matrix (dorsal aspect or top view). f, P. triassica, counterpart with raised veins (ventral aspect or underside view). g, P. triassica under low angle lighting with venation appearing black (x 5.4). h, Monocrinopollis walkeri Comet, round variety (25 x 24 microns; Richmond basin, VB4).
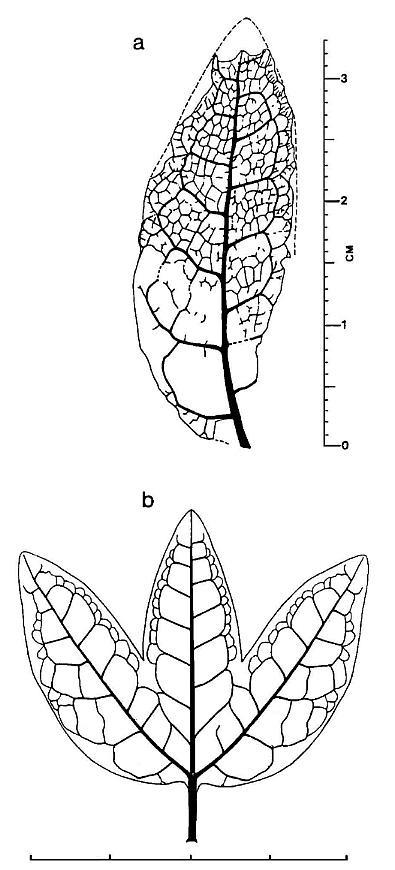
Figure 4 a, Pannaulika triassica nov, gen, et sp., tracing of venation with reconstructed margins and apex (dashed lines). The lower right side of specimen is shown incomplete, because the asymmetrical development of the leaf implies that it's original shape was not entire or simple. Therefore, the dashed line on the right side is not drawn down to the base of the specimen (scale in millimeters and centimeters). b, Reconstruction of what Pannaulika may have looked like if the specimen in Figure 3a represents a lateral lobe of a three lobed leaf (see text for discussion; scale in centimeters).
Diagnosis
Small leaf, at least 3.2 cm in length and 1.3 cm in width, with entire margin and brochidodromous secondary veins arising excurrently from primary and joining subsequent secondary veins at right angles; tertiary veins form at least one row of loops or arches outside secondary loops, creating a festooned brochidodromous vein pattern; tertiary veins also form subaxially- oriented transverse veins within some of the extended secondary loops; very regular, oriented quarternary reticulate venation developed inside secondary and tertiary loops or arches; a band of dendritic or branching quarternary venation extends from tertiary loops or reticulum to the margin of leaf. Secocondary and tertiary arches asymmetrically developed along the side of the leaf with and in the region of a wider lamina; a double row of tertiary arches appears to be developed along part of this expanded margin. Primary vein curves gently in the lower half of the leaf towards the narrower side or opposite the side with the expanded lamina and arches.
Etymology
triassica, for the age of the species.
..
Remarks
The small size of the leaf makes recognition of four vein orders difficult without the aid of enlargement. Vein widths were measured using a binocular microscope with eyepiece micrometer scale (Figure 5). The central primary vein (first order) and brochidodromous (looped or interconnected) secondary veins (second order) both show a gradual tapering of width with distance from theit origin. There is little overlap in widths, and the most common widths (represented by the tallest bars) are quite distinct or separated, as should be expected. The tertiary veins (third order), however, overlap considerably the width range for secondary veins (second order) and reticulate quarternary veins (fourth order). The position and/or disposition of the tertiary veins set them apart from the other veins, and their mean size falls near the overlap between secondary and quarternary veins. The fact that there is so much overlap in size with the secondaries, and that tertiary loops enclose the quarternary retictulum, indicate that they are not part of. the quarternary vein system, while the significant overlap with the widest quarternaries confirms their status as a separate vein system from the secondaries. The recognition of four vein orders helps to distinguish this leaf from ferns and most gymnosperms.
The apex of the leaf is incomplete and eroded, which is indicated by the abrupt termination of primary and secondary veins at the eroded margin; the last right secondary vein (cf. Figure 4a) can be seen to be frayed at the end under high magnification, while a small distal portion of the existing primary vein is missing. The apex appears to. have tapered rapidly to its termination, which is implied by the apparent rapid decrease in width of the primary vein at the preserved apex (Figures 5; 3e). The lateral margin of the leaf is incomplete or eroded in several places, but appears to be almost complete,or natural where a band of branching quarternaries intersects tbe margin outside the tertiary vein arches (Figures 3d; 4a). The quarternary or highest order net venation is very ordered with quadrangular, pentagonal, and hexagonal areoles predominating. Some veinlets can be observed in some of the areoles, but they are usually not as visible as the enclosing. net (Figure 3d,g).
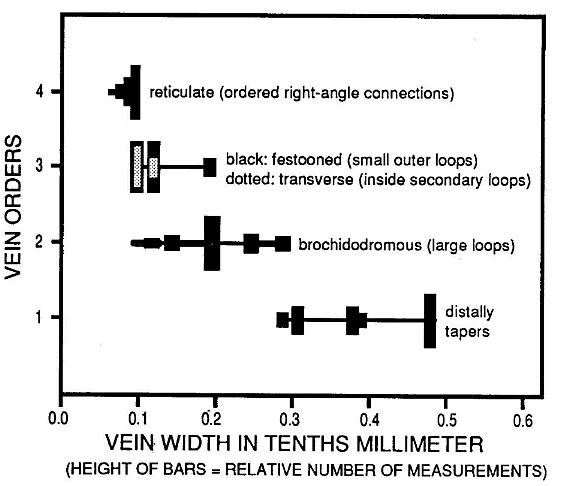
Figure 5. Distribution of vein widths for each of four vein orders. The distinctiveness of the third vein order based on widths and relative position on the leaf serves to distinguish this taxon from fern pinnules and pinnae of comparable size.
The lower lamina on the left side of the primary vein in Figure 4a (underside view, based on raised impression of veins only on that side of specimen) is naturally wider than that on the right side. This fact is supported by the size and shape of the preserved secondary arches, which show transverse tertiary veins present within two of the arches on the left side, but not on the right side, and by the presence of additional tertiary loops outside the row of tertiary loops on the left side. The missing lower right margin appears to have resulted from a diagonal tear that began midway down the right side of the leaf. The primary vein curves gently to the right in the lower half of the leaf. The asymmetrical development or shape of the leaf, along with the curvature of the primary and missing lower right margin, imply that this specimen may be a fragment of a larger leaf. If the fossil represents the lateral lobe of a three-lobed leaf (reconstructed in Figure 4b), the expanded lower left margin, curved primary vein, and nature of the torn right margin make morphological sense. The original size and shape of the leaf are critical in determining systematic affinity, because certain groups of pteridophytes and seed plants can be ruled out if Pannaulika was originally three-lobed and had palmate (actinodromous) venation with three primary veins instead of pinnate venation with only one primary vein.
These deposits and their abundant fauna and flora are described in several papers (Olsen and Gore, 1989; Cornet and Olsen, 1990; Olsen et al., 1978; Olsen, 1979), some of which illustrate the spectacular preservation of insects and aquatic reptiles. However, leaves, seeds, fish, and other invertebrates are equally well preserved despite the high thermal maturity of the sediments (pyrolysis Tmax for whole-rock samples exceeds 400oC; lower Cow Branch Formation Re = 2.15; coals are semianthracite (Robbins, 1982). Leaf fossils are typically preserved as silver-looking compressions on a black matrix. Leaf venation, however, is very well preserved because of the preferential preservation of vascular (lignitic) material, which survives palynological preparation in good condition (Robbins, 1982). Dark brown, highly altered late Carnian-age palynomorphs have been recovered from these lake beds (Robbins and Traverse, 1980; Robbins, 1982). Leaf cuticles are marginally preserved, and it is possible that some may be studied with the use of SEM and transfer techniques.
FLORAL COMPOSITION.
..
The Solite quarry is located near the western side of the Dan River/Danville basin (Figure
1), the deepest side of the half graben. Recently the floras from two different style lake
cycles were studied in detail in order to elucidate the ecological origin of the
dicot-like leaf. There are twelve climatically controlled, lacustrine to playa to
floodplain sedimentary lake cycles [i.e., Van Houten cycles (Olsen and Gore, 1989)]
exposed in the quarry. Whereas the highest four-meter-thick lake cycle (B-12), which
produced the dicot-like leaf, is asymmetrically developed with the deepest water
microlaminated ciaystone facies situated at the base of division 2 (Figure 2b) (Olsen and
Gore, 1989; Olsen et al., 1978), and lowest lake cycle (B-1) is more symmetrically
developed, with the microlaminated portion situated 19 centimeters above the base of
division 2.
The fossil floras of the two lake cycles, although similar qualitatively, are very different quantitatively. Insects are commonly preserved in the microlaminated claystone portions of both cycles. Whereas the B-1 lake flora is strongly dominated by conifer shoots belonging to Pagiophyllum diffusum, P. simpsonii, and other less common Pagiophyllum and Brachyphyllum species (54% versus 9% cycadophyte leaves), the B-12 lake flora is strongly dominated by isolated cycadophyte pinnae and fragmentary fronds belonging to Zamites powellii and Pterophyllum / Pseudoctensis spp. (61 % versus 2% conifer shoots). Leuthardtia ovans, a probable bennettitalean microsporophyll originally described from the Late Triassic of Neuewelt near Basel, Switzerland (Crane, 1986), is relatively common in the B-12 lake flora, and represents the first North American record of this taxon. Winged conifer seeds are much more common in lake B-1, while non-winged cycad and bennettltafean seeds are much more common in lake B-12, reflecting the dominant type of vegetation around the two lakes. As the lakes shrank with climatic deterioration and increasing aridity towards the end of each climatic cycle, pagiophyllous and brachyphyllous conifers returned to dominate the vegetation in the basin. Casts of in situ tree stumps surrounded by fallen conifer shoots appear in the fluvial section at the top of cycle B-12 (Olsen et al., 1978).
The differences in floras and lake cycle symrrtetry imply differences in the rate at which the two lakes developed and the extent to which they developed (i.e., water depth). Lake B-1 apparently grew slowly in size and never completely filled the basin, allowing pre-existing conifer-dominated forests within the basin to persist around the lake margin. Lake B-12, however, appears to have grown much larger and deeper, based on the earlier appearance of a deep lake facies. Because conifer habitats in the basin were mostly eliminated, cycadophyte-dominated communities on the slopes of the mountains and uplands around the basin margin became the dominant lake-side vegetation by default. These mountains may have had as much as one to two kilometers of relief relative to the rift valley floor, similar to that of mountains and uplands bordering East African rift basins today (World Geographic Reference System, 1981).
At the beginning of lake cycle B-12 the upland floral community was the most diverse before basin flooding reduced that diversity. In addition to the Cycadales and Bennettitales, the fossil flora also contains minor percentages of pteriqophytes, sphenophytes (e.g., Neocalamites), lycopods (e.g., Grammaephloios), conifers (e.g., Podozamites and Pagiophyllum), ginkgophytes (e.g., Baiera), non-cycadeoid pteridosperms (e.g., cf. Sagenopteris), and non-bennettitalean anthophytes (e.g., Pelourdea and Fraxinopsis cf. Dechellyia). Interestingly, large tree fern fronds (e.g., Cyathoforma sp.) are present (begin) 5-6 cm above the base of division 2, implying that they may be immigrants after the monsoons intensified. It may be signicant that the dicot-like leaf comes from the lowest part of the microlaminated claystone at the base of cycle B-12, implying that this plant taxon was part of a cycadophyte-dominated community that lived on the drier mountain slopes adjacent to the basin.
COMPARISONS WITH PTERIDOPHYTES
Although the specimen of Pannaulika is probably part of a larger leaf, its asymmetry, constricted base, four orders of venation, and curved primary vein makes its comparison with a pinna of a pinnately compound fern frond untenable. Fern fronds can have more than four orders of venation, but those that do are much larger than the fossil. In those dipteridaceous and polypodiaceous fern taxa with superficial resemblance to the fossil, no more than three vein orders were found in pinnules or those portions of pinnae that compare with Pannaulika in size. The fossil could belong to a palmately lobed or compound frond comparable to Cheiropleuria bicuspis or Bolbitis oligarchia, but again its asymmetry, venation, and curved primary distinguish this leaf from such fern fronds. In addition, the absence of sori or sporangia limit comparison to sterile fern fronds or to ferns with dimorphic leaves.
Among fossil and living ferns, the fronds of the Dipteridaceae most
closely resemble angiosperms, and on occasion have been mistaken for angiosperm leaves
because of their reticulate venation (e.g., Hausmania: Jurassic), secondary veins
that sometimes form weak brochidodromous arches, and pinnae that are fused to various
extents (Meyen, 1987). Such Late Triassic and Early Jurassic dipteridaceous ferns as Clathropteris
and Dictyophyllum were common in the Newark Supergroup, but their venation,
although close; is no more a match for Pannaulika than it is for angiosperms as a
whole. In addition, leaf or frond size is usually several orders of magnitude larger than Pannaulika,
including the larger reconstructed version in Figure 4b. There are other fern taxa
from the early Mesozoic that have reticulate venation, such as Cynepteris (which
resembles extant Woodwardia) (Ash, 1969), Pekinopteris (Hope and Patterson,
1970), and Scoresbya (Harris, 1933), but their venation does not resemble that of Pannaulika,
and their fronds are pinnately compound or much larger than
the dicot-like leaf.
Among living ferns with reticulate venation, such as Woodwardia, Onoclea, Paraleptochilus, and Polypodium, there is again only a superficial resemblance due to the reticulate pattern. Pinna shape, size, and secondary venation are not close. Pantropical and neotropical ferns, such as Ophioglossum reticulatum, Bolbitis nicotianifolia, B. oligarchica, Tectaria heracleifolia, T. incisa, Microgramma lycopodioides, and M. baldwinii and similar in one or more characters with Pannaulika (B. Stolze, pers. comm., 1991), but the differences are equally significant: minor veins tend to parallel the secondaries, areoles closest to the midveins are largest, the reticulations tend not to have right-angle connctions, loop-connected secondaries are rare and weakly developed if developed at all, and there are no loop-connected tertiaries outside the secondary veins (pers. obs. and B. Burger, pers. comm., 1991).
COMPARISON WITH PTERIDOSPERMS
There are a number of fos'sil pteridosperms with reticulate or
anastomosing higher order venation, such as Linopteris and Reticulopteris (Carboniferous),
Glossopteris and Gangamopteris (Permian), Gigantopteris (Permian), Lonchopteris
(Carboniferous-Triassic), Marcouia neuropteroids (Triassic), Furcula
granulifera (Triassic), Quervainia groenlandica (Triassic), Mexiglossa varia
(Jurassic), Dictyozamites hawellii (Jurassic), and Sagenopteris (Triassic-Cretaceous)
(Harris, 1933; Ash, 1972; Taylor, 1981; Meyen, 1987; Mamay, 1989). Most of these taxa have
pinnately compound or simple leaves
lacking pinnately arranged lateral veins that could form loops. If the palmately lobed
reconstruction of Pannaulika is correct (Figure 4b), the palmately compound leaf of
Caytonia (Sagenopteris) might be compared, but it lacks pinnate secondary veins.
The pinnate venation of some of the simple Gigantopteris leaves might be similar if
they possessed loop-connected secondary and tertiary veins and were much smaller.
The simple and forked leaves of Furcula granulifera come the
closest (not very close) to Pannaulika in size and venation, but the reticulum is
not as well developed and its secondary veins only occasionally connect to form weak loops
(Harris, 1933). The paired lobes of the forked variation are symmetrically developed,
unlike Pannaulika, which is asymmetrically developed. The secondary veins diverge
excurrently (at near right angles) from the primary in both Furcula and Pannaulika.
Because excurrent secondaries (instead of decurrent secondaries) in Cretaceous to
Recent angiosperms are considered derived or apomorphic (Hickey, pers. comm., 1991), the
presence of a similar condition in Pannaulika might not be a valid concern if
angiosperms had a pteridosperm ancestry.
COMPARISON WITH ANTHOPHYTES
The anthophytes as currently defined include the Bennettitales, Gnetales, Pentoxylales, and angiosperms. The pinnately compound and simple leaves of the Bennettitales and the simple leaves of Pentoxylon do not compare with Pannaulika. Among the Gnetales only the leaves of extant Gnetum compare with the fossil, although Drewria potomacensis from the Early Cretaceous has reticulate venation, but also parallel venation similar to the cotyledons of extant Welwitschia (Crane and Upchurch, 1987). Yabeiella, a narrow (6-8 cm long), simple pinnately-veined bennettitalean leaf from the Middle Triassic of Argentina, has a distinctive marginal vein, dichotomous to reticulate secondary veins, a primary vein composed of multiple vascular strands, and an apical tooth in some species (Jain and Delevoryas, 1967; Taylor and Hickey, 1992). Although Yabeiella compares with Taylor and Hickey's (Taylor and Hickey, 1992) concept of a pre-angiosperm leaf, it is much less evolved than Pannaulika. Gnetum and Pannaulika share pinnate secondary veins, festooned brochidodromous secondary and tertiary veins, and higher order reticulate venation, but the arches and loops in Gnetum are not as distinct as in the fossil, and the reticulum is not as ordered. These may be insignificant differences considering the variation in these characters among angiosperms. However, the leaves of Gnetum are simple, entire, and symmetrically organized, unlike the fossil. Although the megafossil record for the Gnetales is poor even when including problematic taxa (Table 1), no living or fossil gnetalean has lobate or dissected leaves, although Dechellyia's leafy shoot resembles a compound cycadophyte frond. Furthermore, only living representatives (i.e., the most evolved) are known to possess convergent angiosperm-like characters in their leaves.
All of the features and characters of the fossil can be found among the angiosperms, although no fossil or extant angiosperm leaf is an exact match for the fossil. The rigid box-like gestalt of the brochidodromous arches (imparted by the excurrent secondary veins) and the well-ordered reticulate venation seem too evolved for a leaf at feast 110 million years older than the oldest accepted Cretaceous angiosperm leaf. On the one hand, the fern-like origination of the secondary veins may be a character holdover from a pteridosperm ancestry. On the other hand, the small size, the rigid look of the secondary arches, and the apparent tightness of the lower order venation could be an adaptation for resistance to physical damage from aridity in a monsoon climate, high insolation due to a southerly exposure and poor canopy development in a cycadophyte-dominated community, and wind stress at higher elevations, features which were lost or relaxed among lowland understory Cretaceous angiosperms living in less stressful, wetter environments (i.e., more equable).
According to Hickey and Doyle's (Hickey and Doyle, 1977) definition for leaf rank and advancement during the Early Cretaceous, the least specialized rank one leaves are 5 centimeters or less in length, lack leaf teeth, have a poorly differentiated petiole and blade, are pinriately veined with secondary venation brochidodromous in two or more orders, i.e. festooned, and have a simple, elliptic to ovate or obovate shape. The type of leaf base and petiole cannot be determined for Pannaulika, but all other leaf characters fall within their rank one category with the possible exception of basic leaf shape and primary venation. If Pannaulika was a mulitlobed palmately-veined leaf as the reconstruction in Figure 4b implies, the leaf would be classified no lower than rank two. Yet, some zone one leaf types in the Potomac Group which are classified as rank one have palmate-like venation, such as Vitiphyllum and Proteaephyllum, and are lobate, such as Vitiphyllum (Hickey and Doyle, 1977; Doyle and Hickey, 1976). Thus, there is some uncertainty as to what constitutes a primitive angiosperm leaf, and it may be premature to evaluate Pannaulika based on a scheme for the Cretaceous that has not been adequately tested.
More recently, Taylor and Hickey (1992) presented evidence for an herbaceous origin of angiosperms, with the nonmagnolialean Chloranthaceae and Piperaceae at the bottom of the angiosperm phylogenetic "tree". They hypothesized that the pre- or proto-angiosperm was a diminutive, rhizomatous to scrambling perennial herb, with small simple flowers and simple leaves with reticulate venation. The basal venation of the leaf lamina consisted of at least two proximally diverging pairs of tertiary veins that were free in the midrib and had a different course and behaviour from the distal secondaries (cf. Yabeiella). These basal veins produced an indifferently pinnate to palmate venation, and created a vein pattern that could be ancestral to a palmately lobed leaf with three primary veins.
This hypothesis for pre-angiosperm leaf architecture is important, because one criticism (Hickey, pers. comm., 1991) has been that the primary vein of Pannaulika lacks multiple vascular bundles, as is characteristic of rank one angiosperm leaves (Taylor and Hickey, 1992). Primary veins of palmately veined or lobed leaves that are derived from the basal tertiary veins can lack the multi strand characteristic of the central primary, because they are derived from veins that are not obviously multistrand. If the specimen of Pannaulika triassica represents a lateral lobe, it does not have to have a multistrand primary, particularly for a leaf as small as the holotype.
A second criticism (Hickey, pers. comm., 1991) has been that the tertiary reticulum is too ordered for a primitive angiosperm leaf, and that ths aerioles appear to be mostly complete (usually a derived character). Leaf size has to be taken into consideration when evaluating this character, because even in extant angiosperm leaves with predominantly incomplete aerioles, the aerioles become more ordered and complete in the leaf apex where there is significant constraint placed on vein size and shape. Pannaulika might best be compared with that portion of modern leaves of the same size, i.e. The distal or apical parts of the leaf under 1.3 cm in width.
Pannaulika triassica Cornet, n. gen. et sp. has a very angiosperm-like (dicot-like) appearance. It certainly stands apart from all known Triassic leaf types, and is the first pre-Cretaceous record of a leaf with festooned brochidodromous secondary and tertiary venation and an ordered reticulate quarternary venation with predominantly right-angle connections. This latter character is not present in Gnetum leaves (Burger, pers. comm., 1991), while the combination of these characters may be a synapomorphy for angiospermous. However, it must be pointed out that associated or attached angiospermous reproductive structures are required in order to establish with certainty a relationship with angiosperms. Without such structures an anthophyte affinity is perhaps the finest level of taxonomic resolution that can be attained, but with a strong bias towards angiosperms (i.e., stem angiophyte sensu Doyle and Donoghue, pers. comm., 1992).
Two small reproductive axes and an achene were discovered in the same layer that produced the dicot-like leaf (Figure 6). These specimens are unusual when compared to known Triassic and Jurassic reproguctive structures in that they resemble those of angiosperms. One resembles an aroid spike (monocot-like), while the other is a receptacle bearing two carpel-like megasporophylls (dicot-like). The spike (Figure 6a) is only 3.3 cm long, possesses at least 25 sessile cupule or carpel-like bodies crowded along-the distal 2.2 cm of the axis, and has a near basal cluster of at least three, 7.0 mm long narrow bracts. The cupuie-like bodies are 1.5-2.0 mm in diameter, shaped like an egg or erlenmeyer flask with a blunt to slightly indented apex, and contain, clustered near their bases, impressions of five to twelve tiny ovule-like bodies. It is unlike any known gnetalean or bennettitalean reproductive structures, which possess cupules normally containing but one ovule. It is unlike Caytonia or Lepidostrobus, which have pedicelate and reflexed or fluted cupules.
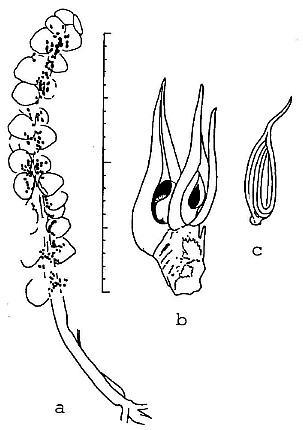
Figure 6 Camera lucida drawings of reproductive structures associated with Pannaulika. a, monocot-like spike; superposition of carpel-like cupules, compression and/or rupture during sedimentary compaction, and poor preservation may account ior the apparent distribution of some ovule-like bodies outside of cupules. b, dicot-like receptacle with two carpel-like megasporophylls, each containing one orthotropous ovule (discovered by B. Axsmith). c, dispersed achene containing one ribbed seed; note similarity in shape to carpels on receptacle (see text for description; scale in millimeters and centimeters).
The dicot-like reproductive structure (discovered by Brian Axsmith) is a receptacle, 5.0 mm long and 3.5 mm wide, bearing two large carpel-like megasporophylls at its blunt apex (Figure 6b). Three or four successive helical scars can be seen along the lateral margin of the receptacle. The ascidate carpels measure 11-13 mm long, and 3-4.5 mm wide at their widest dimension, respectively. They have slightly constricted bases. Attached to the receptacle, adjacent to the carpel on the right, is a 10 mm long, 0.8 mm wide filamentous appendage that resembles a staminodia. Next to it and below it on the receptacle is the base of a narrower filamentous appendage. The carpels are distinctive in that each possesses a 5-6 mm long, 0.8-1.0 mm wide extended stigma/style-like structure, and a suture that runs the entire length of the carpel to the tip of the stigma. The sutures are open on the enlarged ovary, and are spread apart by as much as 2-3 mm; within each carpel a single orthotropous ovule can be discerned. The carpels are at different stages of development, one being larger than the other and containing a larger ovule/seed. The smaller ovule measures 1.8 mm long and 0.6 mm wide.
From the same layer an isolated achene was found that closely resembles the immature carpels in size and shape (Figure 6c). The achene has an elongate stigma/style-like structure identical to that of the carpels, an oval body or ovary, and a small constricted base or pedicel. An impression of a single seed measuring 6 mm long and 2.5 mm wide with longitudinal (vertical) ribbing can be seen inside. But the suture still appears to be open and filled or closed by the seed.
CONCLUSIONS
The association of such reproductive structures with Pannaulika raises the distinct possibility that angiophytes or very primitive angiosperms existed by the Carnian. The open suture is a condition thought to be plesiomorphic, and occurs in some primitive living dicots and palms (e.g., Degeneria, Drimys, and Chamaerops) (Eames, 1961). Carpels with solitary orthotropous ovules are also known in the Amborellaceae, Chloranthaceae, Piperceae, and Saururaceae, considered to be some of the most primitive living angiosperms (Endress, 1990; Taylor, 1991). A manuscript by Axsmith's request on these reproductive structures is in preparation.
It may be significant that diverse and relatively common (1-2%) angiosperm-like pollen (Figures 3a-3c, and 3h) belonging to the Crinopolles Group have been described from slightly older Carnian deposits of the Richmond and Taylorsville basins (located to the east of the Dan River/Danville basin) (Cornet and Olsen, 1990; Cornet;1989a; Doyle and Hotton, 1991). The appearance of the first rift basin environments in the center of Pangaea was soon followed by an almost explosive evolution and diversification of angiosperm-like pollen in the early Carnian (Cornet, 1989a). As climatic conditions changed in the rift zone during the middle Carnian, many early crinopolles pollen types became extinct, while others became more widespread in the rift basins, with the highest.percentages (5-7%) of Monocrinopollis mulleri Cornet and M. microreticulatus Cornet (Figure 3c) recorded near the end of the middle Carnian in cores from along the western margin of the Taylorsville basin, and also in the middle Pekin Formation on the western side of the Deep River basin, N.C. [the cycadophyte-dominated "Hope and Delevoryas" plant locality (Hope and Patterson, 1969)].
It may also be significant that two recent cladistic studies
independently recognized that, as sister groups, the Bennettitales, Gnetales,
Pentoxylales, and angiosperms had a common ancestry no younger than the oldest recognized
member(s) of that group (Doyle and Donoghue, 1986; Crane, 1985); Since
representatives of the Bennettitales and Gnetales are known from the Carnian (Crane, 1985;
Hope and Patterson, 1969; Cornet, 1989b), a separate and distinct phylogenetic lineage
leading to angiosperms has to be at least as old. Recent discoveries of well preserved
leaves and reproductive structures of the enigmatic Late Triassic (Carnian) plant, Sanmiguelia
lewisii, have brought it back into the anthophyte clade (Cornet, 1989b; Cornet, 1986),
and demonstrated a much closer relationship with angiosperms than previously thought
(Hughes, 1976 versus Crane, 1988; Foster and Gifford, 1990). The discovery of Stellatopollis,
Clavatipollenites, and Multimarginites in the marine Oxfordian (early Late Jurassic)
of Normandy, France, now extends the record of Cretaceous and Tertiary-type angiosperm
pollen well before the Cretaceous radiation (Cornet and Habib, 1992). Axelrod presented a
theory in 1952 that attempted to explain the poor pre-Cretaceous angiosperm fossil record
and apparent rapid diversification in the Early Cretaceous. He proposed a tropical upland
origin as far back as the Permo-Triassic and a restriction of these early angiosperms to
the tropics until their migration and radiation into extratropical Cretaceous lowlands.
His theory was never widely accepted. One has to wonder if Axelrod's theory (1952) was not
just the vision of an inquiring naturalist, but a prophecy as well.
I.
Acknowledgements
I thank Prof. P. E. Olsen and D. V. Kent for support via a research
grant to them from the National Science Foundation. I also thank D. I. Axelrod, B.
Axsmith, W. C. Burger, N. Fraser, L. J. Hickey, B. Stolze, J. T. Mickel, and D. W. Taylor
for comments and assistance in evaluating Pannaulika, and P. E. Olsen for use of
his diagrams in Figure 2.
..
..
References
Ash, S..R. (1969) Ferns from the Chinle Formation (Upper Triassic) in
the Fort Wingate area, New Mexico. U.S.
Geol. Surv, Prof Paper 613-D, 51 p.
Ash, S. R. (1972a) Marcouia, gen. novo, a problematical pmnt from the Late Triassic
of the southwestern U.S.A.
Palaeont., 15, 423~29.
Ash, S. R. (1972b) Late Triassic plants from the Chinle Formation in north-eastern
Arizona. Palaeont., 15, 598-618.
Ash, S. R. (1987) Growth habit and systematics of the Upper Triassic plant Pelourdea
poleoensis, southwestern U.S.A.
Rev. Palaeobot. Palynol., 51, 37-49.
Axelrod, D. I. (1952) A theory of angiosperm evolution. Evol., 6,
29-60.
Cornet, B. (1986) The reproductive structures and leaf venation of a Late Triassic
angiosperm. Evol. Theory, 7, 231-309.
Cornet, B. (1989a) Late Triassic angiosperm-like pollen from the Richmond rift basin of
Virginia, U.S.A. Palaeontogr.,
213B, 37-87.
Cornet, B. (1989b) The reproductive.morphology and biology of Sanmiguelia Lewisii, and
its bearing on angiosperm
evolution in the Late Triassic. Evol. Trends in Plants, 3,
25-51.
Cornet, B. and Habib, D. (1992)" AngIosperm-like pollen'from the ammonite-dated
Oxfordian (Upper Jurassic) of France.
Rev. Palaeobot. Palynol., 71, 269-294.
Cornet, B. and Olsen, Po, E. (1985) A summary of the biostratigraphy of the Newark
Supergroup of eastern North
America with comments on Early Mesozoic provinciality, 67-81. In
Weber, R. (ed.): Simposio sobre floras del Triasico
Tardio, su fitogeografia y paleoecologia, Memoria. Instituto de
Geologia Universidad Nacional Autonoma de Mexico.
Cornet B. and Olsen P. E. (1990) Early to .middle Carnian (Triassic) flora and fauna of
the Richmond and Taylorsville basins,
Virginia and Maryland, U.S.A: Guidebook No. I, Virginia Museum of
Natural History, Martinsville, 83 p.
Crane; P. R. (1985) Phylogenetic analysis of seed plants and the origin of angiosperms. Ann.
Missouri Bot. Gard., 72,
716-793.
Crane, P. R. (1986) The morphology and relationships of Bennettitales. In Spicer, R. A.
and Thomas B. A. (eds.):
Systematic and taxonomic approaches in palaeobotany. Systematics
Assoc. Spec. Vol 31, 163-175. Clarendon Press,
Oxford.
Crane, P. R. (1988) Review of Cornet, B., The leaf venation and reproductive structures of
a Late Triassic angiosperm,
Sanmiguelia lewisii. Taxon, 36, 778-779.
Crane, P. R, and Upchurch, Jr., G. R. (1987) Drewria potomacensis gen. et sp. nov.,
an Early Cretaceous member of
Gnetales from the Potomac Group of Virginia. Amer. J. Bot., 74,
1722-1736.
Daugherty, L. H. (1941) The Upper Triassic flora of Arizona. Contributions to
Paleontology, Carnegie lnstitution of
Washington Pub. 256, W.D.C., 108 p.
Doyle, J. A. and Donoghue, M. J. (1986) Seed plant phylogeny and the origin of
angiosperms: an experimental cladistic
approach. Bot. Rev., 52, 321-431.
Doyle, J. A. and Hickey, L. J. (1976) Pollen and leaves from the mid-Cretaceous Potomac
Group and their bearing on early
angiosperm evolution, 139-206. In Beck, C. B. (ed.): Origin and
early evolution of angiosperms. Columbia Univ. Press,
New York.
Doyle, J. A. and Hotton, C. L. (1991) Diversification of early angiosperm pollen in a
cladistic context, 169-195. In
Blackmore, S. and Barnes, S. H. (eds.): Pollen and Spores.
Systematics Assoc. Spec. Vol. 44. Clarendon Press, Oxford.
Eames, A. J. (1961) Morphology of the angiosperms. McGraw-HilI Book Company, New York.
Endress, P. K. (1990) Evolution of reproductive structures and functions in primitive
angiosperms (Magnoliidae). Mem.
N. Y. Bot. Gard., 55, 5-34.
Foster, A. S. and Gifford Jr., E. M. (1990) Comparative morphology of vascular plants,
third edition. W. H. Freeman and
Company, New York.
Goeppert, H. R. and Menge, A. (1883) Die Flora des Bernsteins und ihre Beziehungen zur
flora der Tertiärformation und
der Gegenwart, I. Naturforsch. Ges. Danzig, 63 p.
Harris, T. M. (1933) The fossil flora of Scoresby Sound East Greenland. Medd. Om Gronl.,
85(3),-112 p. Hickey; L. J.
and Doyle, J. A. (1977) Early Cretaceous fossil evidence for
angiosperm evolution. Bot. Rev., 43, 3-104.
Hope, R. C. and Patterson III, O. F., (1969) Triassic flora from the Deep River basin,
North Carolina. North Carolina
Dept. Conserv. Develop., Div. Min. Res. Spec. Pub. 2, 23 p.
Hope, R: C. and Patterson III, O. F. (1970) Pekinopteris auriculata: a new plant
from the North Carolina Triassic. J.
Paleont., 44, 1137-1139.
Hughes, N. F. (1976) Palaeobiology of angiosperm origins. Cambridge Univ. Press, London,
242 p.
Jain, R. K. and Delevoryas, T. (1967) A Middle Triassic flora from the Cacheuta Formation,
Minas de Petroleo,
Argentina. Palaeont., 10, 564-589.
Krassilov, V. A. (1986) New floral structure from the Lower Cretaceous of Lake Baikal
area. Rev. Palaeobot. Palynol.,
47, 9-16.
Krassilov, V. A. and Ash, S. R. {. (1988) On Dinophyton - protognetalean Mesozoic
plant. Palaeontogr., 208B,
33-38.
Krassilov, V. S. and Bugdaeva, E. V. (1988) Gnetalean plants in the
Jurassic of Ust-Balej, East Siberia. Rev. Palaeobot.
Palynol., 53, 359-374.
Mamay, S. H. (1989) Evolsonia, a new genus of gigantopteridaceae from the Lower
Permian Vale Formation, north-
central Texas. Amer. J. Bot., 76,
1299-1321.
Manspeizer, W. and Cousminer, H. L. (1988) Late Triassin-Early Jurassic synrift basins of
the U.S. Atlantic margin, 197-216.
In Sheridan, R. E. and Grow, J. A. (eds.): The Atlantic
continental margin: U.S. The Geological Society of America.
Meyen, S. V. (1987) Fundamentals of palaeobotany. Chapman and Hall, London.
Olsen, P. E. (1979) A new aquatic eosuchian from the Newark Supergroup (Late
Triassic-Early Jurassic) of North
Carolina and Virginia. Pastilla, 176,
1-14.
Olsen, P. E. (1986) A 40 million-year lake record of Early Mesozoic orgital climatic
forcing. Science, 244,789-912.
Olsen, P. E. (1990) Tectonic, climatic, and biotic modulation of lacustrine
ecosystems-examples from Newark Supergroup
of eastern North America, 209-224. In Katz, B. (ed.): Lacustrine
exploration: case studies and modern analogs. Amer.
Assoc. Petro Geol. Memoir 50.
Olsen, P. E. (1991) Field guide to Triassic, for workshop on early Mesozoic small
tetrapods, May 13-18, Fort Royal,
Virginia.
Olsen, P. E. and Gore, P. J. W. (eds.) (1989) Field guide to the tectonics, stratigraphy,
sedimentology, and paleontology
of the Newark Supergroup, eastern North America. Field Trip
Guidebook T351, American Geophys. Union, W.D.C.,
174 p.
Olsen, P. E., Remington, C. L., Cornet, B. and Thomson, K. S. (1979) Cyclic change in Late
Triassic lacustrine
communities. Science, 211, 730-733.
Robbins, E. I. (1982) "Fossil lake Danville": the paleoecology of the Late
Triassic ecosystem on the North Carolina-
Virginia border. Ph.D. Dissertation, The Pennsylvania State
University.
Robbins, E. I. and Traverse, A. (1980) Degraded palynomorphs from the Dan River (North
Carolina)-Danville (Virginia)
basin, 1-11. In Price Jr., V., Thayer, P. A. and Ranson, W. A.
(eds.): Geologic investigations of Piedmont and Triassic
rocks, central North Carolina and Virginia. Carolina Geological
Society Field Trip Guidebook.
Schweitzer, H.-J. (1977) Die Räto-Jurassischen Floren des Iran und Afghanistans.
4. Die Riitische switterblute lrania
Hermaphroditica novo sp. und ihre bedeutung fur die
phylogenie der angiospermen. Palaeontogr., 161B, 98-145.
Taylor, T. N. (1981) Paleobotany, an introduction to fossil plant biology. McGraw-Hill
Inc., New York. Taylor, D. W.
(1991) Angiosperm ovules and carpels: their characters and
polarities, distribution in basal clades, and structural evolution.
Postilla, 208, 1-40.
Taylor, D. W. and Hickey, L. J. (1991) Phylogenetic evidence for the herbaceous origin of
angiosperms. Pl. Syst. Evol.,
180, 137-156.
van Konijnenburg-van Cittert, J. H. A. (1992) An enigmatic Liassic microsporophyll,
yielding Ephedripites pollen. Rev.
Palaeobot. Palynol., 71, 239-254.
Witte, W. K., Kent, D. V. and Olsen P. E. (1991) Magnetostratigraphy and paleomagnetic
poles from Late Triassic-earliest
Jurassic strata of the Newark basin. Geol. Soc. Amer. Bull., 103,
1648-1662.
World Geographic Reference System, Map Series ONC, Sheet M-4, Edition 3, Defense Mapping
Agency Aerospace
Center, Missouri (1981).
Reprints available upon request:
Bruce Cornet
27 Tower Hill Ave.
Red Bank, NJ 07701
USA
This web page was created on 9 June 2002; last updated on 11/12/2002.